Decoding Toxoplasmosis: A Comprehensive Guide of Understanding the Disease (Malaria’s Neglected Cousin)
Dr. Omer Mohi U Din Sofi*
Dr. Idrees Mehraj Allaie*
* Sher-e-Kashmir University of Agricultural Sciences and Technology of Kashmir (SKUAST-K) Srinagar, Jammu and Kashmir, India, 190025.
“No need to be panic, Toxoplasmosis is not any new disease, we have lived with it through century”
HISTORICAL BACKGROUND
Nicolle and Manceaux (1908) discovered a protozoan in the tissues of a hamster-like rodent, the gundi (Ctenodactylus gundi), which was being used in leishmaniasis research at Charles Nicolle’s laboratory at the Pasteur Institute in Tunis. Initially, Nicolle mistook the parasite for a piroplasm and later for Leishmania, but eventually recognized it as a new organism, naming it Toxoplasma gondii based on its shape (toxo = arc or bow, plasma = life) and its host. Similarly, Splendore (1908) identified the same parasite in a rabbit in Brazil, also misclassifying it as Leishmania, but did not give it a name. Over the following 30 years, T. gondii-like organisms were discovered in various other hosts, particularly birds. However, viable T. gondii was first isolated by Sabin and Olitsky (1937), who confirmed it to be identical to the human strain using cross-protection studies. It was later understood that protection against T. gondii involved both innate and specific immune responses. In the 1940s, researchers found that humoral antibodies could eliminate extracellular tachyzoites, but not intracellular ones.
PARASITE MORPHOLOGY
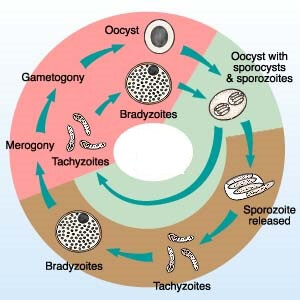
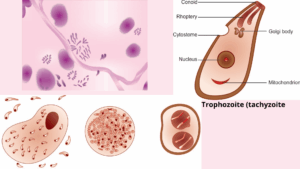
Tachyzoites:The tachyzoite is lunate and is thestage that Nicolle and Manceaux (1908) found in the gundi. Thisstage has also been called a trophozoite, the proliferative form, thefeeding form, and the endozoite. It divides into two by a specialized process called endodyogeny.
Bradyzoite and tissue cysts:The term ‘‘bradyzoite’’ (brady = slow) was proposed by Frenkel (1973) to describe thestage encysted in tissues. Bradyzoites are also called cystozoites.Dubey and Beattie (1988) proposed that cysts should be calledtissue cysts to avoid confusion with oocysts and pseudocysts. Jacobs,Remington, and Melton (1960) first provided a biologicalcharacterization of cysts when they found that the cyst wall wasdestroyed by pepsin or trypsin, but the cystic organisms were resistantto digestion by gastric juices (pepsin–HCl), whereastachyzoites were destroyed immediately. Thus, tissue cysts wereshown to be important in the life cycle of T. gondii because carnivoroushosts can become infected by ingesting infected meat.Cats shed oocysts with a short prepatent period (3–10days) after ingesting tissue cysts or bradyzoites, whereas afterthey ingested tachyzoites or oocysts the prepatent period is longer (≥18 days), irrespective of the number of organisms inthe inocula. Prepatent periods of 11–17 days are thought to result fromthe ingestion of transitional stages between tachyzoite and bradyzoite.
DISEASE TRANSMISSION
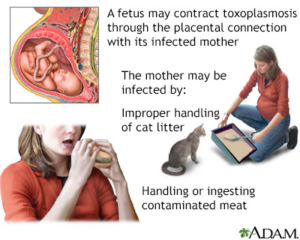
Congenital:The mechanism of transmission of T. gondii remained a mystery until its life cycle was discovered in 1970. Initially transmission by arthropodswas suspected, but this was never proven.Congenital T. gondii infection in a human child was initiallydescribed by Wolf, Cowen, and Page (1939) and later found tooccur in many species of animals, particularly sheep, goats, androdents.
Carnivorism: Congenital transmission occurs too rarely to explainwidespread infection in man and animals worldwide. Weinmanand Chandler (1954) suggested that transmission mightoccur through the ingestion of undercooked meat. Jacobs et al.(1960) provided evidence to support this idea by demonstratingthe resistance to proteolytic enzymes of T. gondii derived fromcysts. They found that the cyst wall was immediately dissolved bysuch enzymes but the released bradyzoites survived long enoughto infect the host. Because the prevalence of T. gondii is much higher insheep than in horses or cattle this illustrated the importance ofcarnivorism in transmission of T. gondii. Epidemiological evidenceindicated it is common in humans in some localities whereraw meat is routinely eaten.
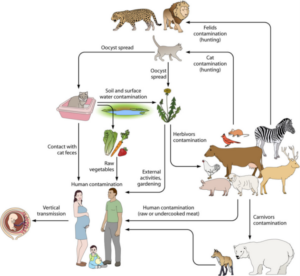
Fecal–oral: While congenital transmission and carnivorism can explain some of the transmission of T. gondii it does not explainthe widespread infection in vegetarians and herbivores.Hutchison (1965), a biologist at Strathclyde University in Glasgow,first discovered T. gondii infectivity associated with cat feces.Vaccination of cats with a live mutantstrain of T. gondii on eight pig farms in the United States reducedthe transmission of T. gondii infection in mice and pigs (Mateus-Pinilla et al. 1999), thus supporting the role of the cat in naturaltransmission of T. gondii.Although T. gondii can be transmitted in several ways, it hasadapted to be transmitted most efficiently by carnivorism in thecat and by the fecal–oral (oocysts) route in other hosts. Pigs andmice (and presumably humans) can be infected by ingesting evenone oocyst (Dubey et al. 1996), whereas 100 oocysts may not infectcats (Dubey 2006). Cats can shed millions of oocysts afteringesting only one bradyzoite, while ingestion of 100 bradyzoitesmay not infect mice orally (Dubey 2006).
TOXOPLASMOSIS IN HUMANS
Congenital toxoplasmosis:Three pathologists, Wolf, Cowen,and Paige from New York, USA first conclusively identifiedT. gondii in an infant girl who was delivered full term by Caesareansection on May, 1938 at Babies Hospital, New York. The girl developed convulsive seizures at 3days of age and lesions were noted in the maculae of both eyesthrough an ophthalmoscope. She died when a month old and anautopsy was performed. At post mortem, brain, spinal cord, andright eye were removed for examination. Free and intracellularT. gondii were found in lesions of encephalomyelitis and retinitisof the girl and viable T. gondii was isolated in animals inoculatedwith tissues from the girl. Sabin (1942) summarized all that wasknown of congenital toxoplasmosis in 1942 and proposed typicalclinical signs of congenital toxoplasmosis: hydrocephalus or microcephalus, intracerebral calcification, and chorioretinitis.
Acquired toxoplasmosis: Sabin (1941) reported toxoplasmosisin a 6-y-old boy from Cincinnati. An asymptomatic childwith initials of R.H. in his name was hit with a baseball bat on October 22,1937. He developed a headache 2 days later and convulsions theday after. He was admitted to the hospital on the seventh day butwithout obvious clinical signs. Except for lymphadenopathy andenlarged spleen, nothing abnormal was found. He then developedneurological signs and died on the 30th day of illness. The brainand spinal cord were removed for histopathological examinationand bioassay. Because of the suspicion of polio virus infection ahomogenate of cerebral cortex was inoculated into mice. Toxoplasmagondii was isolated from the inoculated mice and this isolatewas given the initials of the child and became the famous RH strain. Only small lesions of non-suppurative encephalitis werefound microscopically in the brain of this child without any calcification.This child most likely had acquired T. gondii infectionrecently and the blow to the head was coincidental and unrelatedto the onset of symptoms. This case is historically interestingbecause the RH strain of T. gondii isolated from this boy has since1938 been passaged in mice in many laboratories worldwide.A full appreciationof the clinical symptoms of acquired toxoplasmosis wasachieved when outbreaks of acute toxoplasmosis were reported inadults in the United States.
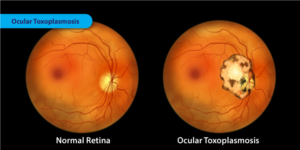
Ocular disease: Before 1950, virtually all cases of oculartoxoplasmosis were considered to result from congenital transmission. Wilder (1952) first identified T. gondii inhistological sections of eyes that had been enucleated. A group of ophthalmologists from southern Brazil initially discovered oculartoxoplasmosis in siblings. Among patients with postnatally acquiredtoxoplamosis who did not have retinochoidal scars before,8.3% developed retinal lesions during a 7-year follow up (Silveiraet al. 1988; Holland 2003). Ocular toxoplasmosis was diagnosedin 20 of 95 patients with acute toxoplasmosis associated with theCanadian waterborne outbreak of toxoplasmosis in 1995 (Burnettet al. 1998).
PATHOPHYSIOLOGY
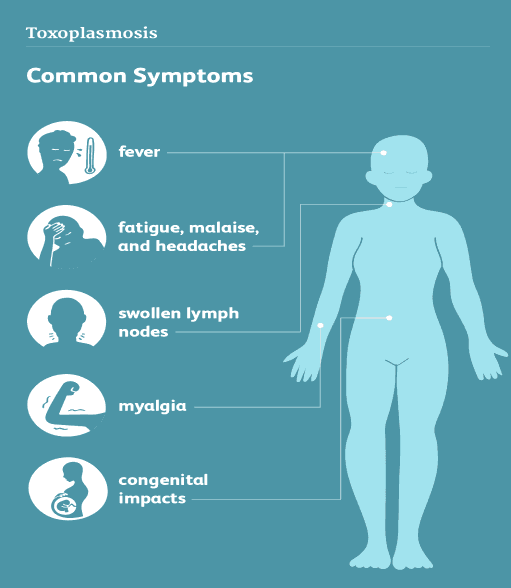
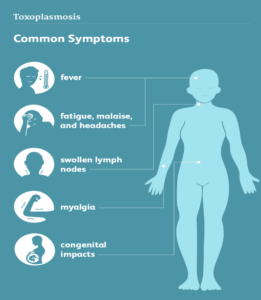
T gondiican overcome the host defenses and disseminate through the body to the liver and spleen, eluding theimmune system and reaching the central nervous system by crossing the highly regulated blood-brain barrier andinfecting the brain, where the parasite resides. Although most infections withT gondiiare subclinical, a smallpercentage of immunocompetent people can develop acute toxoplasmosis that can manifest with nonspecific,constitutional symptoms, including fever, malaise, and lymphadenopathy, as well as central nervous system pathologyand ocular disease—usually posterior uveitis.After the initial infection,T gondiibecomes latent and persists forlife; the latent tissue cysts can reactivate in profoundly immunosuppressed people, and reactivation can also occurduring transplantation from the donor organ.Intracellular growth of tachyzoites results in direct cytopathic effects, cellular inflammation, and necrosis. Type 1 cell-mediated immunity is required to control acute and chronic infection ofT gondii.Thus, any defects in cell-mediatedimmunity predispose the host to severe manifestations of toxoplasmosis.
In response to the damage caused bytachyzoite entry, the gastrointestinal epithelial cells produce chemokines that act as chemical messengers, resulting inthe recruitment of dendritic cells, macrophages, and neutrophils to the site of damage. The entry of tachyzoites intothese inflammatory cells stimulates the production of interleukin-12 that induces interferon-γ (IFN-γ) synthesis bynatural killer cells and T lymphocytes.Synthesis of IFN-γ is required to control acute and chronic infection. Low CD4 lymphocyte counts in patients withAIDS lead to reduced IFN-γ levels, allowing unchecked multiplication of tachyzoites in acute infection andreactivation of bradyzoites in latent infection. This results in severe disease, manifesting as cerebral and extracerebraltoxoplasmosis. Though cell-mediated immunity plays a crucial role in controlling theT gondiiinfection, humoralimmunity also contributes by synthesizing antibodies, modulating CD4 and CD8 T-cell responses, and amplifyingIFN-γ production.
DIFFERENTIAL DIAGNOSIS
The differential diagnosis for toxoplasmosis may include the following:
- Central nervous system lymphoma
- Metastatic brain lesions
- Progressive multifocal encephalopathy
- Cerebral tuberculosis
- Bacterial or fungal brain abscess
- Cytomegalovirus
- Acute HIV infection
- Herpes simplex encephalitis
PREVALENCE STUDIES IN INDIA AND THE WORLD
Seroprevalence of T. gondii varies indifferent regions of the world and in Asia the seroprevalence ranged from 0.8% in Suwon region, South Korea to 63.9% Babol, Iran (Pappas et al. 2009). Incidence of toxoplasmosis was considered to be very low in India and in Kashmir, as compared to that of Western countries (Mittal et al. 1995). In one national survey in India seroprevalence for Toxo-Antibodies (IgG) by ELISA were found in 24.3% with lowest seroprevalences in the northern parts of India (Dhumne et al. 2007).Various studies showed that prevalence of toxoplasmosis in India is similar to that of other parts of world as those reported in Botswana with a seroprevalence of 11% (Kristiahet al. 2009). In USA, 8.9% of seroprevalence was found in women of child bearing age (Jones et al. 2001).
INDIAN OUTBREAK
A most unusual outbreak of ocular toxoplasmosisoccurred in residents of the South Indian city ofCoimbatore, state of Tamil Nadu. The patientswere 12–73 years old and were examined at the AravindEye Hospital Postgraduate Institute of Ophthalmology.The outbreak peaked in 2004; very few cases of T. gondiiretinitis were seen in this eye clinic in years beforeand after this period. A total of 248 patients had oculardisease. Evidence for the acquired toxoplasmosis was asfollows: (i) most patients had high IgM and IgG antibodies,(ii) the retinal lesions were recently acquired and oldscars were absent, and (iii) lesions were mostly unilateral.Most patients were residents of Coimbatore and itssuburbs with minimal immigration from other regionsof India. Water was considered the source of infectionbecause of the demographics; the short period of the outbreakand patients were mostly unrelated to each otherthus discounting sharing common meat meals. The city was supplied by water from the Siruvani Reservoir and the catchment area is likely to be exposed to felids. A heavy rainfall period preceding the outbreak might havecontributed to the outbreak.
DO WE HAVE TO GET RID OF OUR CAT?
No,you do not have to give up your cat. Owning a cat, does not mean youwill be infected with the parasite.It is unlikely that you would beexposed to the parasite by touchingan infected cat because cats usuallydo not carry the parasite on their fur.In addition, cats kept indoors (thatdo not hunt prey or are not fed rawmeat) are not likely to be infectedwith Toxoplasma. But, if you arepregnant, planning on becomingpregnant, or have a weakenedimmune system, it is important toprotect yourself from infection.
PREVENTIVE MEASURES
Prevention of congenital toxoplasmosis:

This can be done by educating pregnantwomen about the sources of infection to encourage themto change their behavior and thus reduce their risk of T. gondiiinfection (primary prevention). The information should coverproper heating of all meat and avoiding raw meat products,hand hygiene for gardening and other soil contact, washing ofvegetables and fruits consumed raw, and precaution measuresfor cleaning of the cat litter box. Although health educationfor pregnant women has existed for decades, its effectivenesshas not been evaluated adequately. Randomized cohortstudies in which the effect of different health education programson the incidence of T. gondii infections are studied areneeded. Secondary prevention of congenital toxoplasmosis isbased on prenatal or neonatal screening and treatment. InFrance and Austria, prenatal screening consists of a first serologicaltest before or early in pregnancy and serological follow-upfor those who tested negative. In France, when seroconversionis demonstrated, spiramycin treatment is initiated andamniotic fluid is tested to check for transmission to the fetus.If amniotic fluid is positive for T. gondii, spiramycin will be replaced by pyrimethamine-sulfadiazine treatment until delivery.In addition, the fetus is checked by ultrasound or magnetic resonanceimaging, and if severe lesions are demonstrated, thepregnancy may be terminated. The effectiveness of prenatal treatment is the subject of debate and large-scale randomized clinical control studies are lacking. Initially, a large Europeanstudy showed only weak evidence for reduction of mother-to-childtransmission, and no effect on the risk of clinical manifestationsat birth could be demonstrated. Later, a reduction of serious neurological sequelae was demonstrated. Recently,French data demonstrated that mandatory monthly prenatalscreening has decreased transmission rates, and implementationof polymerase chain reaction analysis of amniotic fluid in1995 coincided with improved clinical outcomes at age 3 years. In neonatal screening, as carried out in Poland and somestates in the United States and Brazil, dried blood spots are testedfor anti–T. gondii immunoglobulin M. If initial and confirmatorytest results are positive, treatment is initiated and such achild will be examined regularly. There is variation in therapeuticsused and duration of treatment. Consensus about theusefulness of secondary prevention is still absent.
Prevention of infection in food animals:
Herbivorous animals become infected by ingestion of oocysts whilegrazing, drinking contaminated water, or eating contaminated feedstuff. Omnivorous animals such as pigs can also be infectedthrough consumption of rodents, birds, or meat harboring tissuecysts. Measures to prevent infection of food animals includekeeping the animals indoors; keeping cats away fromfarms, feed, and bedding production and storage; providingclean drinking water and blocking access to surface water; implementingstrict rodent control; and refraining from feedingoffal and raw goat whey. Controlled indoor husbandry has drasticallyreduced the prevalence of T. gondii infection in pigs andis considered an important factor in the decrease of seroprevalencesobserved in human populations. However, thecurrent tendency toward organic pig husbandry is likely to increasethe risk of human infection, as a higher prevalence in organichas been demonstrated. For outdoor-rearedanimals (eg, organic pigs and sheep), it is unlikely that preventionmeasures can substantially reduce the prevalence of infection.For these animals, vaccination would be a more feasibleoption; however, a vaccine aimed at preventing tissue cyst formationis currently not on the market. Toxovax, a vaccine licensedonly for prevention of T. gondii abortions in sheep, isavailable. As this vaccine prevents dissemination of parasitesto the placenta, it is likely to also prevent dissemination to other tissues and thereby reduce tissue cyst development,butthis has not been tested experimentally. Even so, a different typeof vaccine would be favorable, as the current vaccine is based onan attenuated live strain of T. gondii (S48), which limits productionand shelf life, demands cold chain administration, and is potentiallyhazardous to the person administering the vaccine.To increase the feasibility of preventing infection in food animals,screening to identify farms with infected animals can be used to limit the interventions to farms with infectedanimals. This could be useful in pig production, as infectedpigs are usually only found at a limited number of farms,whereas almost all sheep farms have infected animals.
Decontamination of meat:
Several methods can be applied to decontaminate infected meat. Freezing meat at −12°C (a properly functioning home freezer) for 2 days will render tissue cysts nonviable, as can γ-irradiationand high-pressure processing. Consumer acceptance maybe a problem because of actual or perceived effects on color, texture,and taste of the meat. In addition, the use of γ-irradiationand high-pressure processing may be restricted by legislation,and may incur high costs. Again, it is possible to increase theefficiency of decontamination by limiting measures to highriskmeat products. In a quantitative microbial risk assessmentincluding 50 meat products, 9 unheated meat products contributed40% of the predicted T. gondii infections. High-risk meat products could be defined as (1) meat destined for preparationof raw meat products (such as raw sausages, carpaccio,or steak tartare) and products that are more likely to be eatenundercooked (eg, beef steak, lamb chops); (2) meat from animalswith outdoor access; or after implementation of screeningon animal or farm level, (3) meat from animals infected withT. gondii; or (4) meat from animals originating from farmswith a high T. gondii prevalence. These definitions could also becombined—for example, focusing on decontamination of meat tobe eaten raw or partially undercooked from high-risk farms.
Reduction of environmental contamination:
Felids are the only definitive hosts forT. gondii, and primaryinfection results in shedding of millions of unsporulated oocystswithin a period of 2 weeks on average. Oocysts become infectiouswithin 1–5 days, depending on temperature and humidity, and can be dispersed from defecation sites mechanicallyand transported to water by runoff. Infectious oocysts are very resistant to environmental conditions including freezing and can survive up to 54 months in cold water and up to 18 months after deposition in soil. The amount of infectiousoocysts in the environment thus depends on feline populationsize, incidence of T. gondii infection in felines, amount ofoocysts shed by an infected feline, fraction of oocysts that endup in the environment and sporulate, and survival of infectiousoocysts. Possibilities to intervene follow from these factors.To regulate the cat population, cat owners need to have their cats spayed or neutered in a timely manner. In addition, control strategies for stray cat populations must be implemented.Cats become infected mainly through hunting or by eating raw meat. To reduce the number of prey animals caught,the cat can be kept indoors, especially at night, or equippedwith a bell or electronic sonic device, but the effect of thesemeasures on the incidence of T. gondii infection has not beenstudied directly. Vaccination of cats may be a more effective way of reducing oocyst shedding by cats. However, no vaccineis currently commercially available. Experimental vaccination the attenuated T-263 strain demonstrated that oocyst shedding after challenge infection could be prevented in 31 of37 kittensin one of the study. This live vaccine has disadvantages similar tothose described for Toxovax, and development of different vaccinationapproaches taking into account potential coverage ratesshould be considered a research priority. Sufficient vaccinationcoverage may be a challenge; although 85% of Dutch cat ownerswho visited a veterinarian were willing, they may present abiased sample of the total population of cat owners. Vaccination of stray cats is only feasible when a capture program is in place.To prevent oocysts from ending up in the environment, catsshould be stimulated to use the litter box, and specific areas withhigh soil contact for humans (eg, sandboxes and vegetable gardens)should be kept free of cats. Cat litter should be disposed ofwith normal household waste, as this route is most likely to resultin effective heat treatment of oocysts. Disposal for compostingcan lead to contamination of the garden, and oocystsflushed down the toilet may contaminate surface waters.
CDC Report about Toxoplasmosis in USA:
- It is the leading cause of death from foodborne illness in the United States.
- The parasite occurs worldwide and can last for long periods of time (up to a lifetime) in human bodies (and other animals).
- People who are pregnant or people who are immunocompromised (have a weakened immune system) can take steps to reduce their risk of infection due to Toxoplasma infection causing more serious health problems.