Formalin-free preservation techniques: glycerine mounting and silicone casting
Authors: Jyoti Saini1, Amit Singh Vishen1 and Aditya Kumar1
1Department of Veterinary Anatomy
College of Veterinary and Animal Sciences, Rani Lakshmi Bai Central Agricultural University, Datia – 475661
Abstract
Formalin (formaldehyde) is widely used for fixation and long-term specimen preservation but poses health risks and regulatory restrictions. Formalin-free approaches such as glycerine (glycerol) mounting for microscopy and silicone casting (including RTV silicone vascular casts and corrosion casting) for macroscopic 3-D anatomical replicas offer safer, effective alternatives for teaching, museum specimens and many research applications. This paper reviews principles, provides step-by-step protocols for glycerine mounting and silicone casting, discusses strengths/limitations, and presents recommended practices to obtain durable, high-quality preparations without formalin. Key methodological choices and safety considerations are highlighted.
Keywords: formalin-free, glycerine mounting, glycerol mounting, silicone casting, corrosion casting, tissue preservation
Introduction
Formalin has been the historical standard for fixation due to its rapid crosslinking and excellent morphological preservation, but its toxicity, carcinogenicity and strong odor drive the need for alternatives in teaching labs and some research contexts. Practical formalin-free approaches include ethanol/glycerin fixation for whole-specimens, glycerine-based mounting media for slides, and polymer casting techniques (e.g., RTV silicone or resins) to create durable 3-D anatomical casts of hollow spaces (vascular, bronchial, luminal). These methods can preserve morphology adequately for many educational and morphological studies while reducing exposure risks. These specimens can serve as practical teaching tools for veterinary anatomy in undergraduate education and museum displays.
Glycerine (glycerol) mounting: aqueous, refractive-index-matched mounting media that preserve wet mounts and delicate tissues (embryos, insect genitalia, algae, small organisms) without dehydration—useful for microscopy and museum slides where long-term fluorescence preservation is required in some formulations.
Silicone casting / corrosion casting: injection of RTV (room-temperature vulcanizing) silicone or other polymers into vascular/ductal systems followed by tissue maceration produces robust 3-D casts ideal for teaching, morphometry, SEM imaging and museum displays. These casts reproduce lumen architecture with high fidelity.
Materials & Methods
A. Glycerine mount
- Specimen preparation: If fresh, rinse small specimens (e.g., insect parts, algae, embryos) in distilled water. For tissues previously fixed in ethanol or formalin alternatives (e.g., ethanol-glycerin), ensure they are equilibrated in water or low-% glycerol to avoid osmotic shock.
- Clearing (optional): For thicker samples, clearing with gentle agents (glycerol itself, lactic acid briefly, or dilute glycerol/water baths) improves transparency.
- Mounting: In the glycerine mount, three successive changes of pure acetone for dehydration were given for 15, 20 and 20 days, respectively followed by transfer to jar containing pure glycerine for 25 days. Finally, it was given two changes of corn starch powder for 5 days each to remove excess glycerine and make it a dry specimen.
- Sealing (for longer storage): Seal edges with nail varnish, rubber solution, or a mounting cement to slow evaporation. Store horizontally at 2–8°C in the dark for fluorescence applications.
- Notes: Glycerol mounts remain partly aqueous—some formulations (glycerol jelly) solidify for true permanent mounts; otherwise slides are semi-permanent and may require re-sealing over years.
B.Silicone casting (vascular/luminal corrosion casting) protocol — condensed
- Specimen preparation & access: Work on fresh or fresh-frozen specimens. Expose and isolate the main inlet vessel; flush with saline to remove blood clots and air. Clamp or ligate branches not to be injected.
- Mix and load casting medium: Prepare RTV silicone (mix base + catalyst per manufacturer) and, if desired, add contrasting pigment. Work quickly—some silicones begin to set within tens of minutes. Use low-viscosity formulations for microvessels.
- Injection: Slowly inject under controlled pressure (manual syringe or peristaltic pump) until peripheral outlets show filling; ligature peripheral drainage if needed. Avoid overpressure to prevent rupture.
- Curing: Allow the silicone to cure fully (time per product, often 1–24 h) at room temperature.
- Maceration (corrosion): Immerse specimen in maceration solution (e.g., 5–10% KOH) at controlled temperature until soft tissues are removed and the cast remains. Rinse thoroughly and neutralize alkali residues. Alternative enzymatic or dilution approaches reduce damage to delicate casts.
- Finishing: Rinse, dry and, if desired, coat with a consolidant or paint for contrast. Mount on stands for display or image with SEM for microvascular architecture.
Results & quality assessment
Glycerine mount specimenare transparent, absence of air bubbles, preservation of cell detail and stain contrast. For fluorescence, check retention over several days/weeks under dark storage. Silicone casts: complete filling of target lumen, no gross leaks or rupture, fine branching visible down to smallest target vessels (dependent on silicone viscosity and injection technique). Proper maceration should leave a clean cast with minimal tissue residues. Advantages & limitations of Glycerine mounting is it is low toxic, simple, good for wet-mounts and fluorescent samples; reversible (specimens can be reprocessed). And Limitations are like some glycerol mounts are semi-permanent (evaporation over years), may not be suitable for long-term archival museum specimens unless using glycerol jelly or further consolidation. While of Silicone casting is excellent 3-D fidelity, durable, non-toxic after curing, ideal for large-scale teaching models and SEM. Limitations are is destructive to surrounding tissue (macération needed), requires careful handling of chemicals (KOH), and variation in polymer formulations affects penetration into microvasculature.
Safety & ethical considerations
Although these techniques avoid formaldehyde, they often use other hazardous chemicals (ethanol, strong alkalis, solvents, pigments). Work in appropriate PPE, with local exhaust or fume hood for solvent/strong alkali steps, and manage waste per institutional guidelines. Obtain ethical approval for animal/human specimens as required.
Conclusion & recommendations
Formalin-free approaches such as glycerine mounting and silicone/corrosion casting provide practical, safer alternatives for many morphological and educational needs. Select medium viscosity and maceration method according to specimen size and the level of microstructural detail required. For slide work, commercial glycerol mounting media with antifade additives are convenient; for 3-D casting, low-viscosity RTV silicones yield the best microvascular replication when injected under controlled pressure. Future work should compare long-term archival stability across different glycerol formulations and silicone chemistries to optimize museum-grade preservation without formalin.
References
- Ravikumar S. Mounting Media – An overview.[Mounting media review]. (Neurobiology Imaging Facility)
- NJIT STG Lab. Clearing and mounting the STG for microscopy — Mounting in glycerol.(NJIT Research)
- Esteban RJR et al. Corrosion Casting, a Known Technique for the Study and …Int J Morphol. (RTV silicone and polymer casting methods). (International Journal of Morphology)
- Shanthini S. An innovative method in venous coronary cast technique.(Examples of corrosion casting applications). (PMC)
- Hammer N. Ethanol-glycerin fixation with thymol conservation— technical report on formalin alternatives. (PubMed)
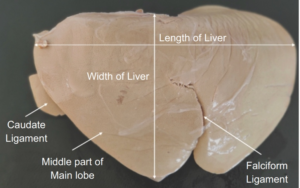 |
| Figure 1. Parietal surface of the liver preserved by glycerine mount technique showing different parts |
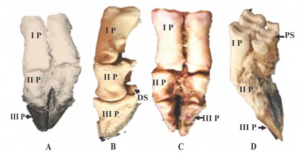 |
| Figure 2. Phalanges of the forelimb of Beetal goat preserved by glycerine mount. A- Dorsal view (I P: 1st phalanx, II P: 2 phalanx, III P: 3 phalanx covered in hoof), B- Lateral view showing 1 , 2 and 3 phalanges nd without hoof, C- Distal of the volar surface of 2 phalanx distal sesamoid (DS) in B and D- Latero-posterior side showing the dorsal view of the proximal sesamoids (PS) which joined back of each condyle large metacarpal
|
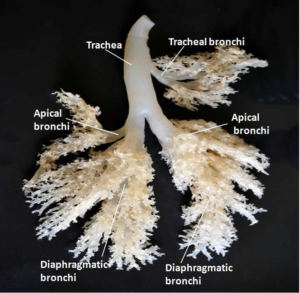 |
| Figure 3: Silicon Cast of Cattle Lung |



