Repeat Breeding in Dairy Cattle: Unravelling the Hidden Barriers to Conception
Sudhanshu Kumar1, Anil Kumar Safi2, Jignesh Vansola3
1Assistant Professor, Department of Veterinary Medicine
2Assistant Professor, Department of Veterinary Anatomy
Mahala Veterinary College, Reengus, RAJUVAS
3Assistant Professor, Department of Animal Nutrition
Mahala Veterinary College, Reengus, RAJUVAS
Corresponding author: anilkumarsafi1501@gmail.com
Abstract
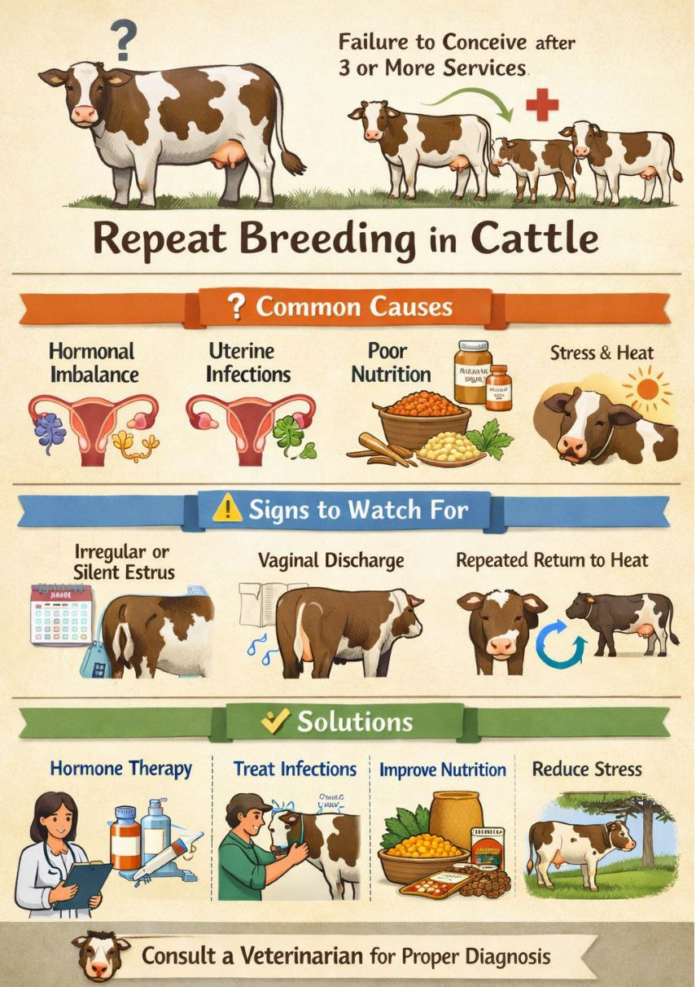
Repeat breeding is a multifactorial reproductive disorder characterized by failure to conceive after three or more regular services in clinically normal cycling cows with no detectable anatomical abnormalities. It represents a major cause of subfertility in dairy herds worldwide, contributing to prolonged calving intervals, increased culling rates and substantial economic losses. The etiology involves complex interactions among endocrine dysfunction, subclinical uterine pathology, nutritional imbalance, improper artificial insemination timing, gamete defects and early embryonic mortality.
Introduction
Efficient reproductive performance is critical for dairy herd profitability. Optimal reproductive efficiency requires calving intervals of approximately 12–13 months. Repeat breeding syndrome significantly compromises this objective. The incidence of repeat breeding varies between 5–30% depending on herd management, environmental conditions, and nutritional status (Ayalon, 1984; Båge et al., 2002).
A repeat breeder cow is generally defined as a cow that:
- Exhibits regular estrous cycles
- Has no palpable or visible abnormalities of the reproductive tract
- Fails to conceive after ≥3 consecutive inseminations
Etiopathogenesis
Repeat breeding is not a single pathological entity but a syndrome with multiple underlying causes.
- Endocrine Dysfunction
Hormonal imbalance plays a pivotal role. Delayed ovulation relative to estrus, inadequate LH surge, luteal insufficiency and suboptimal progesterone concentrations during early diestrus impair fertilization and embryo survival (Wiltbank et al., 2016).
Inadequate pre-ovulatory follicular development may compromise oocyte competence. Low post-ovulatory progesterone is associated with altered endometrial gene expression and reduced embryo survival (Mann & Lamming, 2001).
- Subclinical Endometritis
Subclinical uterine inflammation significantly contributes to repeat breeding. Even in the absence of purulent discharge, polymorphonuclear cell (PMN) infiltration alters uterine receptivity and interferon-τ signaling, impairing maternal recognition of pregnancy (Sheldon et al., 2006).
Endometritis is generally diagnosed by cytological examination when >5–10% neutrophils are detected in endometrial cytological samples beyond 21–28 days of postpartum (Kasimanickam et al., 2004).
- Early Embryonic Mortality
Embryonic losses between days 8–16 post-fertilization are common in repeat breeders. Causes include:
- Inadequate uterine environment
- Insufficient progesterone
- Heat stress
- Genetic abnormalities
Roughly 30–40% of embryos may be lost during early pregnancy in high-producing dairy cows (Diskin & Morris, 2008).
- Oocyte and Gamete Quality
High metabolic stress in early lactation negatively affects oocyte competence. Elevated non-esterified fatty acids (NEFA) and β-hydroxybutyrate (BHBA) alter follicular fluid composition, compromising oocyte maturation (Leroy et al., 2008).
- Nutritional Factors
Negative energy balance delays resumption of ovarian cyclicity and reduces conception rates. Trace mineral deficiencies (Cu, Zn, Se & Mn) and inadequate phosphorus impair reproductive function. Antioxidants like vitamin E and selenium enhance uterine immunity and embryo survival rate (Underwood & Suttle, 1999).
- Heat Stress
Thermal stress disrupts folliculogenesis, steroidogenesis and embryo development. Elevated rectal temperatures (>39.5°C) around insemination reduce conception rates significantly (Hansen, 2009).
- Artificial Insemination Errors
Improper semen storage, thawing technique, insemination timing and deposition site (e.g., cervical instead of uterine body) contribute to fertilization failure.
Diagnostic Approach
A systematic diagnostic protocol includes:
- Detailed reproductive history
- Per rectal and ultrasonographic examination
- Endometrial cytology
- Progesterone profiling
- Body condition scoring
- Evaluation of estrus detection efficiency
- Semen quality assessment
Ultrasonography aids in detecting luteal insufficiency, follicular cysts and early embryonic loss.
Therapeutic Strategies
Treatment depends on the underlying cause.
- Hormonal Therapy
- GnRH at insemination for delayed ovulation
- hCG to support luteal function
- Progesterone supplementation (CIDR) in luteal insufficiency
- Ovsynch and timed AI protocols
Meta-analyses indicate improved conception rates in repeat breeders treated with synchronization protocols (Stevenson et al., 2014).
- Treatment of Subclinical Endometritis
- Intrauterine antibiotics (e.g., cephapirin)
- Prostaglandin F2α in luteal phase
- Improved postpartum management
- Nutritional Correction
- Balanced energy-protein ratio
- Mineral mixture supplementation
- Antioxidant therapy
- Management Interventions
- Accurate heat detection (activity monitors & pedometers)
- Heat stress abatement (cooling systems & shade structures)
- Strict semen handling protocols
Conclusion
Repeat breeding is a multifactorial reproductive disorder involving endocrine, uterine, metabolic, and environmental components. A systematic diagnostic and therapeutic approach targeting underlying causes can significantly improve fertility outcomes. Advances in reproductive endocrinology, ultrasonography and herd management technologies offer promising tools to mitigate this condition.
References
Ayalon, N. (1984). A review of embryonic mortality in cattle. Journal of Reproduction and Fertility, 54, 483–493.
Båge, R., Gustafsson, H., Larsson, B., Forsberg, M., & Rodríguez-Martínez, H. (2002). Repeat breeding in dairy heifers: follicular dynamics and estrous cycle characteristics. Theriogenology, 57, 2257–2269.
De Vries, A. (2006). Economic value of pregnancy in dairy cattle. Journal of Dairy Science, 89, 3876–3885.
Diskin, M.G., & Morris, D.G. (2008). Embryonic and early fetal losses in cattle and other ruminants. Reproduction in Domestic Animals, 43(Suppl. 2), 260–267.
Hansen, P.J. (2009). Effects of heat stress on mammalian reproduction. Philosophical Transactions of the Royal Society B, 364, 3341–3350.
Kasimanickam, R., Duffield, T.F., Foster, R.A.(2004). Endometrial cytology and reproductive performance in dairy cows. Theriogenology, 62, 9–23.
Leroy, J.L.M.R., Vanholder, T., Delanghe, J.R.(2008). Metabolic changes in follicular fluid of dairy cows. Reproduction, 135, 239–250.
Mann, G.E., & Lamming, G.E. (2001). Relationship between maternal endocrine environment and embryo survival. Journal of Reproduction and Fertility, 121, 175–182.
Sheldon, I.M., Lewis, G.S., LeBlanc, S., & Gilbert, R.O. (2006). Defining postpartum uterine disease in cattle. Theriogenology, 65, 1516–1530.
Stevenson, J.S., Pulley, S.L., & Hill, S.L. (2014). Reproductive management of dairy cows using synchronization protocols. Journal of Dairy Science, 97, 3407–3423.
Underwood, E.J., & Suttle, N.F. (1999). The Mineral Nutrition of Livestock. CABI Publishing.
Wiltbank, M.C., Baez, G.M. & Garcia-Guerra, A. (2016). Pivotal periods for pregnancy loss in dairy cattle. Theriogenology, 86, 239–253.