Zoonoses and Climate Change: A Double Threat to Global Health
Alok Kumar Chaudhary PhD ( IVRI)
Assistant Professor
Department of Veterinary Medicine, COVAS, DUVASU,Mathura-281001
Contact: drvetalok@gmail.com
Introduction:
Globally, rise of emerging and re-emerging zoonotic diseases is closely interconnected with climate change, driven by shifts in temperature, precipitation patterns, and ecosystem dynamics. The gravity of this threat is underscored by the collaborative efforts of major international health organizations, including the World Health Organization (WHO), the World Organization for Animal Health (WOAH, formerly OIE), the Food and Agriculture Organization of the United Nations (FAO), the Centers for Disease Control and Prevention (CDC, USA), and the United Nations Environment Programme (UNEP). These agencies jointly support the One Health Commission, which advocates for a collaborative, multisectoral, and transdisciplinary approach to mitigating zoonotic disease threats. Working under the One Health framework, these organizations have identified over 200 known zoonotic diseases, which are responsible for approximately 60% to 70% of all emerging infectious disease cases (Taylor et al 2001). These include viral diseases (e.g., rabies, influenza), bacterial infections (e.g.,brucellosis, leptospirosis), parasitic infections (e.g., toxoplasmosis), and fungal diseases, reflecting the broad and complex nature of zoonotic transmission. A substantial body of scientific evidence indicates that animals are key drivers in the transmission of zoonotic diseases. Wildlife species, along with pets, companion animals, birds, fish, and other aquatic organisms, serve as major reservoirs for zoonotic pathogens accounting for approximately 60% of existing human infectious diseases and nearly 70% of emerging infections in the human population. In addition to vertebrate reservoirs, insects play a significant role as vectors and intermediate hosts in the transmission cycle of various zoonotic pathogens. While insects such as mosquitoes, ticks, and flies are well-established vectors, the increasing entomophagy (consumption of insects) in regions like Asia, Africa, and South America presents a growing concern.


Image 1: Global One Health coordination Image 2: National One Health implementation
Approximately 2 billion people worldwide consume beetles, caterpillars, grasshoppers, and other edible insects as part of their diet, which, despite offering high nutritional value, may serve as potential sources of zoonotic transmission. Edible insects have been found to harbor a range of bacterial, parasitic, and food borne pathogens, thereby posing a potential risk for the emergence of novel zoonotic diseases in human populations. Climatic crises, driven by human activities such as deforestation, fossil fuel use, and unsustainable land practices, lead to rising temperatures, altered rainfall, and extreme weather events. These disruptions increase human-animal interactions and disturb ecological balance, creating ideal conditions for the emergence and spread of zoonotic diseases. Climate change thus serves as a major catalyst for global zoonotic threats. A multidispilnary strategy that takes into account the ecological, social, and environmental elements that influence the genesis and spread of zoonotic diseases is needed to address these interrelated problems under one health mission worldwide.
Overview of Zoonoses: Terminology and Definition
The German physician and pathologist Rudolf Virchow introduced the term ‘Zoonosis’ (Pleural: Zoonoses)” in 1880 to describe human diseases shared with animals (Leal et al 2022). Zoonotic diseases were defined in 1951 by the joint FAO/WHO expert committee on Zoonoses as “diseases and infections that are naturally transmitted between vertebrate animals and man” The term ‘zoonoses’ is derived from the Greek words ‘zoon’ meaning animal, and ‘nosos’ meaning disease. Besides, this term some other terminology are considered that mainly focused on the predominant path of transmission between humans and other vertebrates (Chomel et al, 2009).viz anthropozoonosis” (from animals to humans) and “zooanthroponosis” (from humans to animals) and amphixenoses (transmitted in either direction and maintained in humans and lower vertebrate animals) .
Classification of Zoonotic Diseases: A Concise Overview
Zoonoses are classified based on various factors to understand their origin, transmission, and control strategies. The primary classification systems include:
Classification based on Etiological agent
a) BacterialZoonoses
(e.g. Anthrax, Brucellosis, Plague, Leptospirosis, Salmonellosis, Lyme Disease)
b) ViralZoonoses
(e.g., Rabies, arbovirus infections, KFD, yellow fever, influenza, CCHF)
c) Protozoal zoonoses
(e.g. Toxoplasmosis, Trypanosomiasis, Leishmaniasis)
d) Helminthic zoonoses
(e.g. Echinococcosis (Hydatid Disease), Taeniasis, Schistosomiasis, Dracunculiasis)
e) Fungal zoonoses
(e.g.,Histoplasmosis, Cryptococcosis, Superficial Dermatophytes.)
f) Rickettsial zoonoses
(e.g. murine typhus, tick typhus, scrub typhus, Q-fever)
g) Ectoparasites zoonoses
( e.g. scabies, myiasis)
Classification based on Reservoir Host (WHO/FAO, 1967)
a) Anthropozoonoses: Reservoir Host Animals → Humans (e.g., rabies)
b) Zooanthroponoses: Reservoir Host Humans → Animals (e.g., tuberculosis)
c) Amphixenoses: Bidirectional transmission (e.g., Ringworm)
Classification based on Zoonotic Transmission Cycle (Epidemiological)
a) Orthozoonoses (Direct): Transmitted directly (e.g., rabies, brucellosis)
b) Cyclozoonoses: Require ≥2 vertebrate hosts (e.g., echinococcosis, taeniasis)
c) Pherozoonoses (Metazoonoses): Involve both vertebrates and invertebrate vectors
(e.g., plague, arboviruses)
d) Saprozoonoses: Involve vertebrate hosts and non-living environments
(e.g., mycoses, swine erysipelas)
Classification based on Environmental
a) Synanthropic Zoonoses: Restricted to domestic settings (e.g., listeriosis)
b) Exoanthropic Zoonoses: Circulate in wild animals, transmitted via vectors
(e.g., tick-borne encephalitis)
Classification based geographical
a) Endemic zoonoses(Present in many places and affect many people and animals)
b) Epidemic zoonoses(Sporadic in temporal and spatial distribution)
c) Pandemic zoonoses (worldwide)
d) Emerging and Re-emerging zoonoses
(Newly appearing in a population or have existed previously but are rapidly increasing in incidence or geographical range).
Zoonoses: Global and Indian Public Health Impact
Globally, zoonotic diseases contribute significantly to the burden of illness and mortality. Estimates suggest that nearly one billion cases of zoonotic illnesses and millions of deaths occur each year. Roughly 60% of emerging infectious diseases (EIDs) reported worldwide are of zoonotic origin. In the last 30 years alone, more than 30 novel human pathogens have been identified, with about 75% of them originating from animals. In India, zoonotic diseases remain a critical but underreported public health concern. Historical outbreaks, such as plague, have claimed an estimated 12 million lives since 1898. Rabies continues to be a persistent threat, accounting for approximately 20,000 deaths annually. Over 1.7 million individuals are reportedly bitten by potentially rabid animals each year and seek post-exposure prophylaxis at health centers. Diseases like typhus, which caused widespread fatalities during World War I, and brucellosis, which leads to the loss of around 30 million man-days annually, highlight the socio-economic burden. Brucellosis alone causes an estimated ₹24 crore (INR 240 million) in annual economic losses due to decreased productivity in cattle and buffalo populations. Japanese encephalitis has emerged as another major zoonotic threat, causing frequent outbreaks with significant health consequences.. While Kala-azar (visceral leishmaniasis) is considered zoonotic globally, in India it is traditionally regarded as non-zoonotic, despite epidemiological patterns suggesting otherwise. Conversely, cutaneous leishmaniasis, once thought to be purely human-transmitted (anthroponotic), has now been confirmed as zoonotic in India, with the Indian desert gerbil (Meriones hurrianae) identified as a natural reservoir. India’s unique epidemiological landscapewith over 80% of its population living in rural areas, in close proximity to more than 512 million livestock, 729 million poultry, and large populations of wild and semi-wild animalsamplifies the risk of zoonotic disease transmission. Recently the COVID-19 pandemic, believed to have originated from an animal source, has infected over 700 million people and resulted in nearly 7 million deaths (WHO, 2023). The economic toll exceeds USD 20 trillion, encompassing healthcare costs, loss of productivity, and disrupted global trade (World Bank, IMF).Other examples include: Ebola Virus Disease (2014–2016): Over 11,300 deaths and USD 2.8 billion in economic losses in West Africa.Avian Influenza: Mass culling of poultry has disrupted food security and caused billions in trade losses. These cases underscore the importance of integrated surveillance and response systems. The One Health approach, championed by WHO, FAO, WOAH, and UNEP, emphasizes the need for multisectoral collaboration to address the complex interface between human, animal, and environmental health..
Climate Change: Key Climate Drivers Affecting Health
Climate change, driven primarily by human activities, poses an escalating threat to planetary and public health worldwide. The key global drivers of climate change include: (i) elevated greenhouse gas (GHG) emissions particularly carbon dioxide (CO₂), methane (CH₄), and nitrous oxide (N₂O) largely from fossil fuel combustion, deforestation, and industrial agriculture; (ii) unsustainable energy production and consumption (iii) Rapid urbanization and (iv) land-use changes, including intensive livestock farming and monoculture cropping systems. According to the Intergovernmental Panel on Climate Change (IPCC, AR6, 2023), global average surface temperatures have increased by approximately 1.1 °C since the pre-industrial era (1850–1900) and are projected to surpass 1.5 °C within the next two decades if emission trends continue. This warming has led to rising sea levels, glacial retreat, ocean acidification, and increased frequency of extreme weather events including heat waves, wildfires, floods, and tropical cyclones posing major risks to ecosystems and human populations. About one-third of total anthropogenic GHG emissions are attributed to global food systems, with animal-based food production contributing nearly double the emissions of plant-based systems (Crippa et al., Nature Food, 2021).

Image 3: Key drivers of global health risks.
In the Indian context, the impacts of climate change are particularly severe due to the country’s ecological and socio-economic vulnerabilities. The India Meteorological Department (IMD) reports that the average land surface temperature in India has risen by ~0.7 °C between 1901 and 2018, with 2022 ranking as the fifth warmest year on record, showing a deviation of +0.51 °C from the mean (IMD, 2023). India witnessed over 2,200 extreme weather events in 2022 alone, causing the loss of more than 3,000 human lives, extensive damage to infrastructure, and disruptions in food and water security (MoES, 2023). Additionally, Indian Ocean warming has intensified tropical cyclone activity, while erratic monsoon patterns have resulted in both prolonged droughts and flash floods. Such hydro-climatic extremes elevate the risks of vector-borne diseases (e.g., dengue, chikungunya, and malaria), heat-related illnesses, respiratory distress due to poor air quality, and malnutrition stemming from crop failures and food insecurity. The projected temperature increase of 2–4 °C over the Indian subcontinent by the end of the 21st century further amplifies these concerns (IMD, 2020; IPCC, 2023). Global and regional data underline the urgency of implementing robust climate-resilient health systems, decarbonization of energy sectors, sustainable agriculture, and intersectoral policies based on the One Health framework to protect ecosystems, animals, and human populations
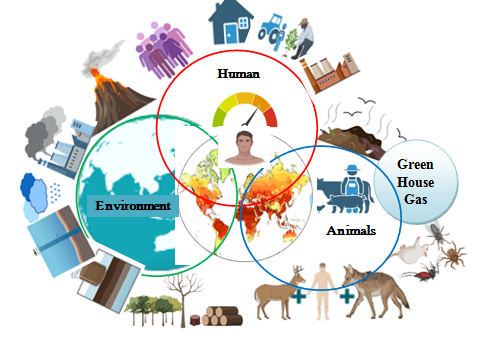
6-Zoonoses and Climate Change: Interconnected Dynamics
The increasing intersection of climate change and zoonotic diseases has emerged as a significant public health concern in the 21st century. Both phenomena are intricately linked through multiple ecological, environmental, and socio-economic pathways. Ecosystem and biodiversity changes brought about by climate change may have an impact on the occurrence and spread of zoonotic illnesses. Vector-borne diseases, foodborne diseases, and waterborne diseases are the major infectious diseases related to climate change. Climate change modifies the conditions under which zoonotic pathogens evolve, survive, and transmit, while anthropogenic pressures such as deforestation and urbanization create new interfaces between humans, domestic animals, and wildlife, accelerating spillover events .The World Health Organization (2021) has estimated that approximately 60% of all emerging infectious diseases are zoonotic, with more than 70% originating from wildlife. Climate change acts as a critical catalyst in this dynamic, enhancing the likelihood, intensity, and geographical spread of zoonotic outbreaks.
Ecological Disruption and Vector Range Expansion
Climate variables such as temperature, humidity, and precipitation directly influence the biology, breeding, and distribution of arthropod vectors, including mosquitoes, ticks, and sandflies. The life cycles of mosquitoes, ticks, and sandflies are dependent on ambient temperature due to their ectothermic life cycles. This impact is significant as over 17 percent of infectious illnesses are transmitted through vectors. For example, rising temperatures reduce the extrinsic incubation period of vector-borne pathogens, increasing the efficiency of disease transmission. Mosquitoes such as Aedes aegypti and Anopheles spp. are now thriving in higher altitudes and latitudes due to warming climates, facilitating the emergence of dengue, Zika, chikungunya, and malaria in previously non-endemic areas (Rocklöv & Dubrow, 2020).According to the IPCC Sixth Assessment Report (2023), global mean temperatures have increased by 1.1 °C since the pre-industrial era, and climate suitability for vectors such as Ixodes scapularis (Lyme disease vector) has significantly increased in North America and parts of Europe. Likewise, tick-borne encephalitis is now endemic in areas where it was previously rare, such as southern Scandinavia and the Baltic region (Semenza & Suk, 2018)
Climate-Driven Spillover Events
Climate change exacerbates ecological fragmentation, habitat degradation, and wildlife migration, all of which increase the frequency of zoonotic spillovers. Extreme weather events particularly droughts and floods disrupt ecosystems and force wildlife into closer contact with human settlements and domestic animals. A case in point is the Nipah virus in Southeast Asia. Originally hosted by fruit bats (Pteropus spp.), the virus spilled over to pigs and subsequently to humans, often during periods of climate-related food scarcity and habitat loss caused by deforestation and agricultural expansion (Pulliam et al., 2012). Similar risks are being observed in tropical Africa and South America, where intensified land-use changes and climate variability create high-risk interfaces for diseases such as Ebola, Lassa fever, and Yellow fever. Airborne diseases, such as influenza, tuberculosis, and aspergillosis, are likewise impacted by climate-induced changes. Hotter, drier summers lead to increased dust particle mobilization, aiding in the long-distance transport of spores and pathogens. Soil disturbance due to construction or drought, combined with wind, facilitates airborne transmission. In urban areas, crowding during seasonal shifts further contributes to respiratory disease outbreaks. Recent studies have also demonstrated increased influenza virus concentrations during dust storm events. Moreover, newly emerging zoonotic viruses, including MERS-CoV and Coxiella, could evolve into potential pandemics, particularly as their distribution shifts into previously unaffected regions due to changing climatic conditions.
Flooding drive Waterborne Zoonoses,Rodent-Borne and Post-Monsoon Risks
Heavy rainfall and flooding driven by erratic monsoonal patterns significantly increase the incidence of waterborne and rodent-borne zoonotic disease. In countries like India, post-monsoon outbreaks of leptospirosis have become increasingly common. Leptospira spp. bacteria thrive in waterlogged areas contaminated with the urine of infected rodents or domestic animals. According to the India Meteorological Department (IMD, 2023), the intensity and frequency of extreme rainfall events have increased, with the country recording over 2,200 extreme weather events in 2022, many of which resulted in flooding. This not only exacerbates disease risk but also damages sanitation infrastructure, leading to the spread of cholera, hepatitis A, and cryptosporidiosis, especially in informal urban settlements. Warming sea surface temperatures also enhance the proliferation of Vibrio cholerae, especially in coastal and estuarine areas. Studies have demonstrated increasing cholera outbreaks in Bangladesh and parts of East Africa during El Niño periods, where ocean warming correlates with heightened disease activity (Baker-Austin et al., 2013).
Food borne Pathogens and Climate Variables
Climate change also influences foodborne zoonoses. Higher ambient temperatures, coupled with increased humidity, promote the survival and multiplication of bacterial pathogens such as Salmonella spp., E. coli, and Vibrio spp. Temperature increases of just 1 °C have been linked to 5–10% rises in salmonellosis incidence in Europe. Flooding of croplands can also introduce microbial contaminants into the food chain. Additionally, trematode infections may rise due to the enhanced spread of freshwater snails in expanding warm habitats.

Image -4: Zoonoses and Climate Change: Interconnected Dynamics
Wildlife Habitat Loss and Human Encroachment
Deforestation, mining, and agricultural expansion frequently exacerbated by climate-induced land degradation displace wildlife and shrink natural habitats. As a result, animals are forced into closer proximity to human populations, increasing the risk of zoonotic spillover. For example, the Amazon basin, a hotspot of biodiversity, has seen extensive deforestation driven by cattle ranching and soybean cultivation. These activities, coupled with warming trends, disturb the balance of local ecosystems and increase the emergence risk of diseases such as leishmaniasis and hantavirus. In India, forest cover degradation in the Western Ghats and northeastern states has increased human-wildlife conflict and zoonotic disease emergence, including Kyasanur Forest Disease (KFD), a tick-borne viral hemorrhagic fever. The IPBES Global Assessment (2019) highlights that over one million animal and plant species are threatened with extinction, largely due to anthropogenic activity, and this biodiversity loss severely impairs natural ecosystem functions, including disease regulation.
Climate Change and Zoonoses: Human Behavioral Dimensions
Human behavior plays a critical intermediary role in the interaction between climate change and zoonotic disease transmission. As climate parameters—such as temperature, humidity, and precipitation—continue to fluctuate, they drive shifts in agricultural practices, land use, urban migration, and water management strategies. These shifts not only modify ecological landscapes but also influence human exposure to animal reservoirs and vectors, thereby altering zoonotic risk profiles (El-Sayed & Awad, 2018).
a) Migration, Urbanization, and Ecosystem Disruption
Climate-induced droughts, floods, and heatwaves can lead to large-scale displacement of both human and livestock populations. The movement of people and animals from environmentally stressed regions into urban and peri-urban areas creates densely populated habitats where sanitation may be poor and human-animal contact frequent—conditions conducive to zoonotic spillover. According to global assessments, human activities have significantly modified 75% of terrestrial ecosystems, 65% of marine environments, and 85% of wetlands, contributing to habitat loss and fragmentation (IPBES, 2019). These changes disturb the natural geographic distribution of wild animal populations and disease vectors. Rodents, for example, are particularly resilient to such disturbances due to their high fecundity, adaptability, and mobility. Moderate levels of disturbance may even enhance rodent population growth, thereby increasing their role as reservoirs for pathogens such as Leptospira, Hantavirus, and Yersinia pestis (Meerburg et al., 2009). However, severe habitat destruction can disrupt rodent population dynamics and force vectors and hosts into new, sometimes more human-populated areas.
b) Livestock Management and Animal Husbandry Practices
Climate change is also influencing livestock production systems. Rising temperatures and frequent heat stress events have led to the adoption of indoor confinement systems in animal farming. While this can reduce heat exposure for livestock, it also increases the potential for pathogen amplification and interspecies transmission due to overcrowding and poor ventilation. Furthermore, the development and use of new livestock breeds more tolerant to heat may inadvertently introduce unfamiliar host-pathogen interactions. Extended grazing seasons and pasture expansion into marginal lands may also increase opportunities for livestock–wildlife interactions, potentially facilitating the spillover of novel pathogens.
c) Changes in Water Use, Hygiene, and Recreation
Warmer climates influence human behavior by increasing the frequency of water contact activities such as bathing, swimming, and irrigation. These behaviors, particularly when combined with poor water sanitation infrastructure, increase the risk of exposure to waterborne zoonotic pathogens such as Vibrio cholerae, Leptospira spp., Schistosoma, and Cryptosporidium. Individuals swimming with open wounds are especially at risk of contracting such infections. In addition, higher ambient temperatures lead to increased water consumption and may reduce hygiene practices due to water scarcity, contributing to outbreaks of diarrheal diseases and other gastrointestinal infections. The indirect effects of climate change on behavior—such as altered dietary habits, outdoor food consumption, and waste handling—also have implications for the spread of food- and environment-borne zoonoses.
Resistance and Pathogen Adaptation
Climate change may also accelerate the evolution and resilience of zoonotic pathogens. Environmental stressors can favor genetic mutations, promoting antimicrobial resistance and making treatment of diseases more difficult. Enhanced survival strategies among vectors and microbes enable them to withstand conventional control measures, thereby sustaining transmission cycles even in adverse conditions.
One Health mission: The 4S Mitigative Framework Against Zoonosis and Climate Change
The One Health approach is an internationally endorsed paradigm that aims to optimize health outcomes for humans, animals, and ecosystems by fostering integrated actions across disciplines. In the context of zoonotic disease emergence and the escalating impacts of climate change, One Health adopts a proactive stance through a 4S framework: Surveillance, Synergy, Strategy, and Sustainability.
Surveillance: Strengthening Early Warning Systems
Integrated surveillance is fundamental to One Health’s core objective. It involves systematic collection, analysis, and interpretation of data from human, animal, and environmental health domains to ensure early detection and control of zoonotic threats. Global platforms like GLEWS (Global Early Warning System) and GOARN (Global Outbreak Alert and Response Network), as well as local digital systems such as AfyaData in Tanzania and PODD in Thailand, have successfully detected abnormal health events and initiated rapid responses. Despite these advances, data fragmentation, especially in low- and middle-income countries, remains a barrier to comprehensive surveillance.
Synergy: Fostering Interdisciplinary Collaboration
Climate-sensitive zoonoses require transdisciplinary solutions. Synergistic collaboration between veterinary medicine, public health, environmental sciences, and data technology is essential for controlling disease outbreaks. For example, the integration of remote sensing and GIS-based Earth observation tools has allowed researchers in the Amazon and Southeast Asia to map high-risk zones of vector-borne diseases like dengue and leishmaniasis. However, gaps remain in integrating behavioral, socio-economic, and cultural dimensions into zoonotic disease modeling, limiting effective policymaking and community participation.
Strategy: Prioritizing Zoonotic Threats
Strategic planning under One Health includes identifying and ranking priority zoonoses using tools like the One Health Zoonotic Disease Prioritization Process (OHZDP). This participatory method fosters intersectoral consensus and enables resource allocation for high-impact diseases. Countries like Ethiopia, India, and Uganda have adopted OHZDP to tailor their public health responses. However, local-level implementation and disease prioritization often lack follow-up funding, operational frameworks, and integration with climate resilience strategies.
Sustainability: Aligning with Climate and Development Goals
Sustainability in the One Health framework underscores long-term resilience building through environmentally sound, economically feasible, and socially inclusive interventions. This involves promoting climate-smart agriculture, minimizing wildlife–human interface, reducing antimicrobial resistance, and aligning with UN Sustainable Development Goals (SDGs 3, 13, 15, and 17). While many policies promote sustainability in silos, cross-sectoral enforcement remains weak, and environmental degradation continues to facilitate pathogen spillover and biodiversity loss.
Research Gaps and Challenges
Despite its growing adoption, the One Health framework faces notable scientific and operational gaps: Limited climate–pathogen modeling, especially in data-deficient regions. Inadequate genomic surveillance to detect mutations in zoonotic agents under ecological stress. Weak integration of socio-political and cultural variables in One Health interventions. Poorly established governance and funding mechanisms for sustained collaboration across sectors. These gaps hinder anticipatory and preventive capacity at both national and international levels.
Conclusion
In summary, the 4S model—Surveillance, Synergy, Strategy, and Sustainability—provides a robust, evidence-based mitigation approach under the One Health banner to combat the growing convergence of zoonotic diseases and climate change. Integrating research, policy, and practice through this framework strengthens resilience, responsiveness, and health system preparedness. Addressing the identified gaps through interdisciplinary science, global cooperation, and adaptive governance is imperative. Institutionalizing the One Health approach not only mitigates future pandemics but also safeguards global health, biodiversity, and planetary integrity in an era of environmental uncertainty
References:
- Taylor LH, Latham SM. Woolhouse ME. Risk factors for human disease emergence. Philos. Trans. R. Soc. Lond. B Biol Sci. 2001 Jul; 356(1411):983–989.
- Leal Filho W, Ternova L, Parasnis SA, Kovaleva M, Nagy GJ. Climate Change and Zoonoses: A Review of Concepts, Definitions, and Bibliometrics. Int J Environ Res Public Health. 2022 Jan 14;19(2):893. doi:
- Joint FAO/WHO Expert Committee on Zoonoses. World Health Organization. Food and Agriculture Organization of the United Nations Second Report. WHO IRIS; Proceedings of the Joint WHO/FAO Expert Committee on Zoonoses; Stockholm, Sweden. 11–16 August 1958; [(accessed on 14 July 2021)]
- Chomel B. Encyclopedia of Microbiology. Academic; San Diego, CA, USA: 2009. Zoonoses; pp. 820–829.
- (2023). Sixth Assessment Report (AR6): Synthesis Report.
- India Meteorological Department (IMD). (2023). State of Climate Report – India. Ministry of Earth Sciences.
- Ministry of Earth Sciences (MoES), Government of India. (2023). Annual Climate Summary.
- Crippa, M., Solazzo, E., Guizzardi, D., et al. (2021). “Food systems are responsible for a third of global anthropogenic GHG emissions.” Nature Food, 2(3), 198–209.
- (2022). Climate Change and Health. Fact Sheets.
- (2023). Sixth Assessment Report – Synthesis Report.
- (2022). Climate Change and Health: Key Facts.
- Pulliam, J.R.C. et al. (2012). “Agricultural intensification, priming for persistence and the emergence of Nipah virus: a lethal bat-borne zoonosis.” Journal of the Royal Society Interface, 9(66), 89–101.
- Ogden, N.H. et al. (2014). “Recent and projected future climatic suitability of North America for Ixodes scapularisand Borrelia burgdorferi.” Parasites & Vectors, 7:532.
- Baker-Austin, C. et al. (2013). “Emerging Vibrio risk at high latitudes in response to ocean warming.” Nature Climate Change, 3(1), 73–77.
- (2021). Zoonoses: Managing Public Health Risks at the Human–Animal–Environment Interface.
- India Meteorological Department (IMD). (2023). State of Climate Report – India.